Products
- Titanium Tablet
- Carbide Powder
- Nitride Powders
- Hydride Powders
- Silicide Powders
- Ti5Si3 Titanium Silicide
- TaSi2 Tantalum Silicide
- VSi2 Vanadium Silicide
- CrSi2 Chromium Silicide
- HfSi2 Hafnium Silicide
- Mg2Si Magnesium Silicide
- MnSi Manganese Silicide
- MoSi2 Molybdenum Silicide
- NbSi2 Niobium Silicide
- Ni2Si Nickel Silicide
- TiSi2 Titanium Disilicide
- WSi2 Tungsten Silicide
- FeSi2 Ferro Silicide
- ZrSi2 Zirconium Silicide
- Cu5Si Copper Silicide
- CoSi2 Cobalt Silicide
- Yttrium Disilicide,YSi2
- Calcium Silicide,CaSi2
- Boride Powders
- TiB2 Titanium Boride Powder
- TaB2 Tantalum Boride powder
- ZrB2 Zirconium Boride Powder
- VB2 Vanadium Boride Powder
- CaB6 Calcium Boride Powder
- CrB2 Chromium Boride Powder
- CoB Cobalt Boride Powder
- SiB6 Silicon Boride Powder
- HfB2 Hafnium Boride Powder
- AlB2 Aluminum Boride Powder
- MgB2 Magnesium Boride Powder
- MnB2 Manganese Boride Powder
- MoB2 Molybdenum Boride Powder
- NbB2 Niobium Boride Powder
- Ni2B Nickel Boride Powder
- WB2 Tungsten Boride Powder
- MoB Molybdenum Boride powder
- WB Tungsten Boride powder
- FeB Iron Boride powder
- Metal Alloy Powders
- High purity powders
- Max materials
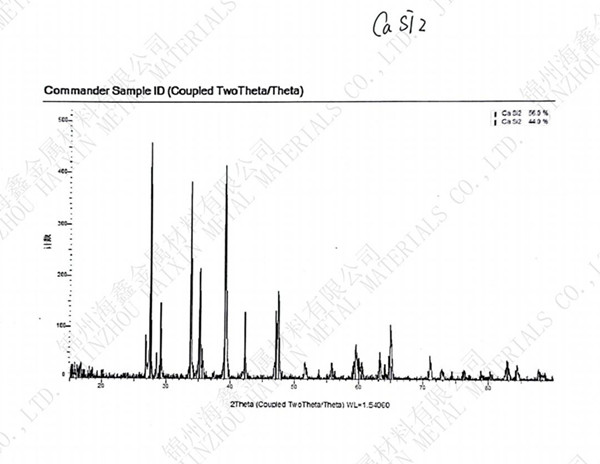
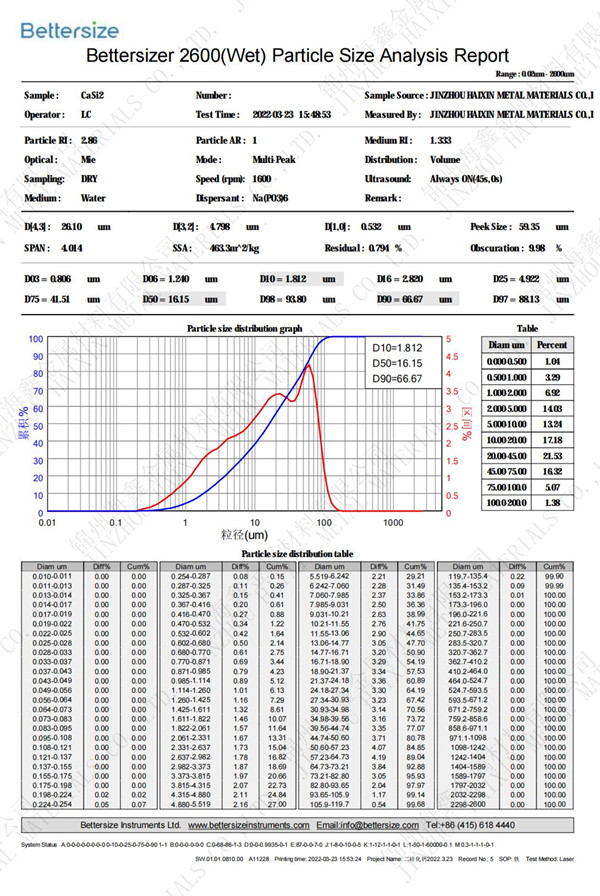
Calcium Silicide,CaSi2
Chemical formula:CaSi2. molecular weight:96.25. Gray brown hexagonal plate-shaped crystal with metallic luster. The relative density was 2.5. It is insoluble in water and soluble in hydrochloric acid to produce siloxane. Preparation method: CASI and Si mixed and heated, mole ratio 1:1 ,or Cao and ......
Send Inquiry
Product Description

Chemical formula:CaSi2.
molecular weight:96.25.
Gray brown hexagonal plate-shaped crystal with metallic luster.
The relative density was 2.5.
It is insoluble in water and soluble in hydrochloric acid to produce siloxane.
Preparation method: CASI and Si mixed and heated, mole ratio 1:1
,or Cao and Si heated at high temperature.
application: making explosives.
CAS: 12013-56-8
Molecular formula: CaSi2
Molecular weight: 96.24900
Melting point: 1020oc
Density: 2.5 g / cm3
Appearance and character: shape: Crystal





Calcium silicide is used for deoxidation mostly used for scientific research.
It is designed to increase the amount of heat conductive material to prevent
the formation of oxidization of iron.. It is used for deoxidation of steelmaking a
nd additive of special steel.
Calcium silicate can react with water to form combustible hydrogen silicide gas.
Therefore, after calcium silicate fire breaks out, it can’t be extinguished with water
or carbon dioxide fire extinguisher, sand or dry powder fire extinguisher can be used.







